STS101 (DHE Nasal Powder)
STS101 combines the Satsuma powder technology with an easy-to-use nasal delivery device to create a reliable and convenient DHE product potentially able to provide the unique clinical advantages of DHE while overcoming the shortcomings of existing DHE products.
STS101 has a number of key advantages that we believe may provide significant benefits over other acute treatments for migraine and result in robust and consistent clinical performance. These advantages arise from our proprietary dry-powder formulation, which incorporates a mucoadhesive drug carrier and engineered drug particle technologies, and our proprietary nasal delivery device.
STS101 is an investigational product for the acute treatment of migraine and is not approved by the U.S. Food and Drug Administration.
We have conducted several Phase 1 clinical trials designed to evaluate the comparative pharmacokinetics, safety and tolerability of STS101 versus liquid nasal spray and injectable DHE products marketed in the United States. In each of our Phase 1 comparative pharmacokinetic trials, DHE exposure (plasma concentration over time expressed as area-under-curve) following STS101 administration at all times exceeded that for Migranal® DHE mesylate liquid nasal spray 2.0 mg (which is currently the only DHE product approved by the U.S. Food and Drug Administration based upon demonstration of efficacy and safety in double-blind, placebo-controlled clinical trials), and was similar to DHE exposures achieved with DHE mesylate intramuscular (IM) injection, which has consistently demonstrated robust efficacy by 2 hours post-administration in controlled clinical trials. In addition, pharmacokinetic variability with STS101 was significantly lower than Migranal.
In our most recent Phase 1 clinical trial conducted in 2021, administration of STS101 5.2 mg resulted in rapid and sustained achievement of target plasma concentrations with low pharmacokinetic variability. STS101 5.2 mg achieved mean DHE plasma concentrations of 2.0 ng/mL within approximately 15 to 20 minutes, and remained above 1.0 ng/mL for 2 hours after administration. Consistent with prior trials, STS101 5.2 mg demonstrated a favorable safety and tolerability profile, with low rates of adverse events observed. We believe this pharmacokinetic profile should translate to rapid and enduring anti-migraine effects with minimal adverse events.1-8
DHE Plasma Concentrations 0-2 hours
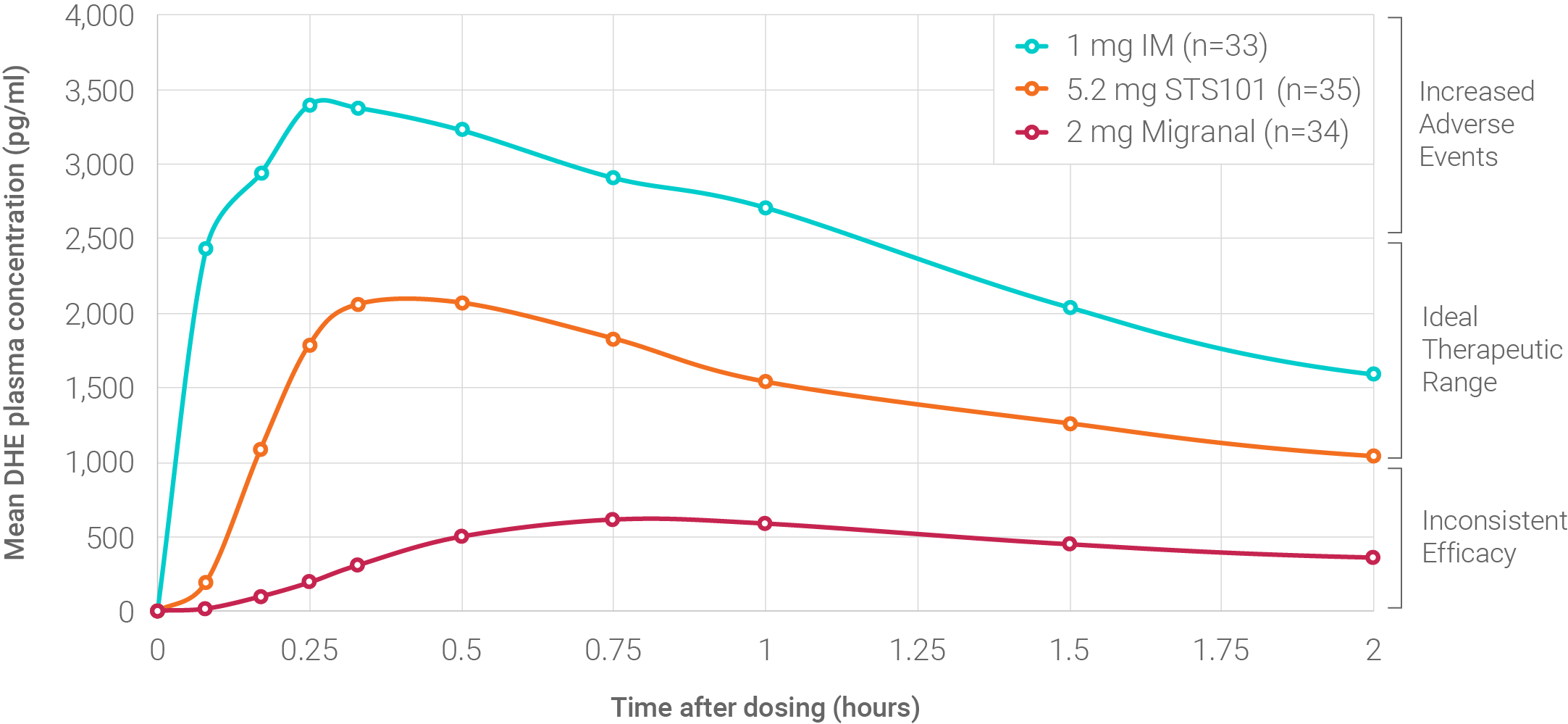
We believe STS101’s unique PK profile has the potential to translate into a differentiated treatment that provides fast onset and robust efficacy by 2 hours post-administration, with low rates of migraine recurrence. Satsuma has completed both the SUMMIT and ASCEND studies supporting the safety and efficacy of STS101 in the acute treatment of migraine.
Click here for more information about our clinical trials.
Advantages of Powder Over Liquids

Less susceptible to dripping out of the nose or down the back of the throat

Adheres to the nasal mucosa, providing better absorption

Better stability versus liquid formulations
Sources
1. Satsuma data on file
2. Humbert, Clin Pharm & Therapeutics 1996
3. Migranal Full Prescribing Information
4. Kellerman, J of Aerosol Medicine & Pulmonary Drug Delivery 2013
5. Aurora. Headache 2009
6. Aurora. Headache 2011
7. Schran, Curr Therapeutic Res 1994
8. Winner, Arch of Neurology 1996
